Polyglutamine (polyQ) diseases are caused by an unstable expansion of genomic cytosine-adenine-guanine (CAG) trinucleotide repeats beyond a specific threshold. In the healthy population, the number of CAG repeats lies between 10 to 51, while 55-87 CAG repeats is associated with the disease. The CAG tracts are translated intoexpanded glutamine regions in the respective proteins, promoting their self-assembly and amyloid structure formation. Within this context, Spinocerebellar ataxia type 1 (SCA1) and SCA3 are inherited neurodegenerative disease belonging to the class of polyglutamine (polyQ) diseases. We apply computational methodologies to elucidate the early stages of protein aggregation mechanism at molecular level. Furthermore, in agreement with the previous experimental findings, our results can be considered as a basis for a future design of ataxin aggregation inhibitors that will require several key conformations identified in the present study as molecular targets for ligand binding.
Publications:
– G. Grasso, U. Morbiducci, J.A. Tuszynski, A. Danani, M.A. Deriu, “Destabilizing the AXH tetramer by intra- and inter-dimer mutations: from molecular mechanism to potential anti-aggregation strategies”, Biophysical Journal, 114 (2), P323-330, 2018, doi10.1016/j.bpj.2017.11.025
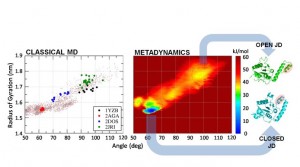
– G. Grasso, J. A. Tuszynski, U. Morbiducci, G. Licandro, A. Danani, M. A. Deriu, “Thermodynamic and Kinetic Stability of the Josephin Domain Closed Arrangement: Evidences from Replica Exchange Molecular Dynamics”, Biology Direct Vol. 12, Issue 2 (2017)
– M. A. Deriu, G. Grasso; J. A. Tuszynski, D. Massai, D. Gallo, U. Morbiducci, A. Danani, “Characterization of the AXH Domain of Ataxin-1 Using Enhanced Sampling and Functional Mode Analysis”, Proteins: Structure, Funtions and Bioinformatics , Vol. 84, Issue 5, 2016, 666–673
– G. Grasso, M. A. Deriu, J. A. Tuszynski, D. Gallo, U. Morbiducci, A. Danani, “Conformational Fluctuations of the AXH monomer of Ataxin 1”, Proteins: Structure, Funtions and Bioinformatics , Vol. 84, Issue 1, January 2016, 52–59